In recent years, major jurisdictions worldwide have been strengthening antitrust regulation of transactions that fall below notification thresholds but they consider may still nevertheless potentially harm competition. By analyzing the competitive concerns and review approaches in two very important cases, this article will explore the recent antitrust enforcement priorities in the semiconductor and pharmaceutical industries and provide corresponding compliance recommendations for industry professionals.
The European Commission and its member state antitrust authorities have been filling the regulatory gap for transactions that fall below notification thresholds but which still may potentially harm competition by introducing or strengthening “call-in powers”[1]. In China, the Anti-Monopoly Law (2022 Amendment) (the “AML”) grants antitrust authority the power to call in transactions that do not meet the notification thresholds but may have anti-competitive effects. Recently, the State Administration for Market Regulation (“SAMR”) announced two cases involving the semiconductor and pharmaceutical industries, which are both key areas of antitrust regulatory focus in China; the SAMR conditionally approved one and prohibited the other. Despite the fact that the transactions did not meet the notification thresholds, the SAMR nonetheless called in the cases, conducted substantive reviews and intervened.
This sends a clear signal that the substantive competitive impact of a transaction has been prioritized by authorities and that there will be tightened regulatory of potential monopolistic behaviors such as unfairly high pricing, refusal to supply, and bundling arrangements.
Please click here to read the PDF version.
Summary of Below-Threshold Cases Raising Competition Concerns
Based on publicly available information, there have been four merger cases in recent years that fell below the notification thresholds but nevertheless raised competitive concerns, primarily in the pharmaceutical and semiconductor industries.
Pharmaceutical industry
For the pharmaceutical industry, the SAMR has consistently regarded it as a key enforcement area and has closely monitored the potential impact of transactions on the industry’s competitive landscape. Due to the highly segmented nature of the pharmaceutical market, a company may possess significant market power or even a dominant market position within a narrowed relevant market, even if its annual revenue falls below the notification thresholds. The Antitrust Guidelines for the Pharmaceutical Field issued in 2025 explicitly note that a company’s annual revenue may not meet the notification thresholds set by the State Council because the market for certain drugs is relatively small or because the company is in an early stage of drug development, such as for innovative drugs. If a merger in the pharmaceutical sector does not meet the notification thresholds but there is evidence that it has or may have the effect of eliminating or restricting competition, the SAMR can require the participating undertakings to submit a notification by issuing a written notice to them.
As early as 2019, Hunan Er-Kang Pharmaceutical’s proposed acquisition of Henan Jiushi Pharmaceutical was voluntarily abandoned by the transaction parties after lengthy discussions with the SAMR. This proposed transaction occurred against the backdrop of the SAMR’s 2018 investigation into chlorphenamine API price manipulation, where both transaction parties were found to be abusing dominant market positions and were penalized. By halting the transaction, the SAMR was essentially preventing the transaction from further reducing market competition.
In 2023, Simcere Pharmaceutical made a hostile takeover bid for Tobishi. Although Simcere Pharmaceutical’s acquisition of equity of Tobishi did not meet the notification threshold, Tobishi proactively submitted a merger notification to the SAMR as a defensive measure against the hostile takeover by Simcere. The SAMR accepted the case and ultimately approved the transaction with restrictive conditions to maintain a stable supply of the relevant API.
In 2025, the SAMR intervened in Wuhan Yongtong’s acquisition of Shandong Huatai Pharmaceutical, ultimately blocking the transaction and ordering a restoration to the state prior to the concentration. This was the fourth prohibited concentration since the implementation of the AML and the first case in the pharmaceutical industry to be prohibited with an order to restore the pre-concentration state (i.e. unwind the transaction).
Semiconductor industry
For the semiconductor industry, a core sector in China’s industrial policy, the Chinese government has consistently emphasized its strategic significance. Accordingly, the SAMR maintains strict scrutiny of transactions in this sector, and several previously approved cases with restrictive conditions have also involved semiconductor companies.
In the case of Synopsys’s proposed acquisition of Ansys, although Ansys’s annual revenue in China did not meet the notification thresholds set by the State Council[2], the SAMR nonetheless exercised its call-in power under Article 26(2)[3] of the AML, requiring a mandatory notification and ultimately granting a conditional clearance (See below for more details). This marks China’s first conditional approval of a below-threshold transaction through call-in power in merger review history and constitutes a notable practical milestone in the implementation of China’s antitrust review system.
How does the Antitrust Authority evaluate whether a transaction has anti-competitive effects?
In assessing horizontal mergers between competitors, antitrust authorities generally assess whether the transaction is likely to eliminate or restrict competition based on multiple factors: the purpose of the transaction (e.g. whether it aims to eliminate competition); the combined market share of the parties in relevant markets post-transaction; changes in market concentration (such as the HHI index); the potential for unilateral effects (such as price increases or quality reductions) and coordinated effects (such as collusion); and potential impacts on innovation and technological progress.
For non-horizontal mergers, such as those involving companies at different levels of the supply chain, antitrust authorities assess potential anti-competitive effects by examining: the purpose of the transaction; its potential to create foreclosure effects (e.g. excluding competitors by controlling essential inputs or access to customers); the possibility of gaining a competitive advantage by accessing sensitive information; the potential for coordinated price increases across the supply chain; the possibility of anti-competitive practices such as tying, refusal to deal, or exclusive arrangements.
The above summary is based on the provisions of the AML, Guidelines for the Review of Horizontal Concentrations of Undertakings, and Guidelines for the Review of Non-Horizontal Concentrations of Undertakings (Draft for Comments), among others.
Conditional Approval of Synopsys’ Share Acquisition over Ansys
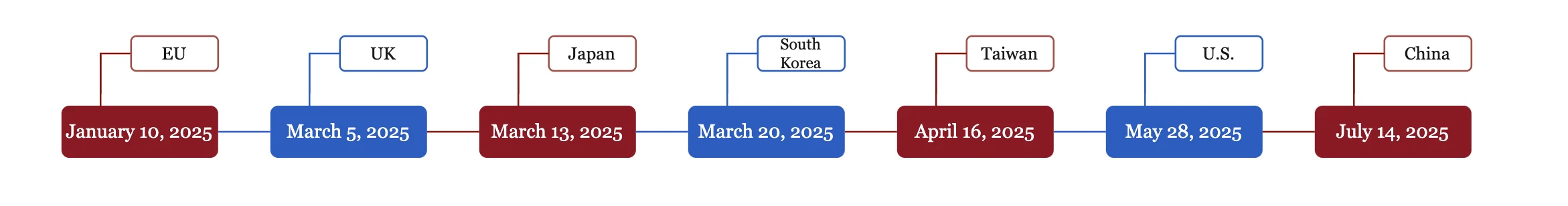
On July 14, 2025, the SAMR officially approved Synopsys’ share acquisition over Ansys with remedial conditions. Since the transaction was first announced on January 16, 2024, it has undergone antitrust reviews in multiple major jurisdictions, including the European Union, the United States, and the United Kingdom. The completion of the review in China marked the final step in the global antitrust review process. Subsequently, Synopsys announced that the transaction was completed on July 17,2025. The following aspects of the case are particularly noteworthy:
Figure 1: Global Antitrust Clearance Timeline for Synopsys’ Share Acquisition of Ansys
The SAMR’s first conditional approval for below-threshold case by exercising call-in power
This transaction marks the first case in China’s antitrust enforcement history in which the SAMR exercised its call-in power to require a notification for an below-threshold merger and grant conditional approval. Moreover, it serves as a landmark application of the “call-in powers” explicitly conferred on regulators under the 2022 amendments to the AML, marking a significant milestone in the law’s implementation. According to the review decision issued by the SAMR, the transaction was notified under the normal case procedure on July 10, 2024, and finally obtained approval on July 14, 2025. The review took 369 days (including 57 days of “stop-the-clock” period). The lengthy review period, which lasted nearly a year, not only reflects the Chinese antitrust authority’s significant concern about the potential competitive effects of the transaction but also underscores its cautious approach toward transactions that, while below the notification thresholds, are industry-sensitive or could have substantial competitive impact.
Key Takeaways
The call-in power is expected to become a key tool for China’s antitrust authority in regulating future below-threshold transactions. A transaction falling below the notification thresholds is not automatically exempted from antitrust scrutiny. During the transaction planning stage, companies must remain highly attentive to their merger control compliance obligations, particularly when the transaction involves market-influential players or operates in sensitive industries. In such cases, a thorough assessment of the potential impacts on China’s competitive landscape and its upstream/downstream enterprises is essential, requiring thorough advance risk evaluation and mitigation planning. Where appropriate, companies may also consider pre-notification consultations with the SAMR regarding the transaction structure, market implications, and feasible remedies. This proactive approach helps mitigate the risk of reactive regulatory intervention, enhances transaction predictability, and should help to avoid adverse consequences arising from uncertainties in antitrust review.
Semiconductors & tech-intensive industries remain a key focus of antitrust regulation
This transaction involves the acquirer, Synopsys, a global leading provider of electronic design automation (“EDA”) software and semiconductor design IP, and the target, Ansys, which primarily focuses on the research, development, and sales of simulation and analysis (“S&A”) software. Ansys’s S&A software is widely used in semiconductor design workflows by chip designers, qualifying it as EDA software from a technical standpoint.
Although EDA software and design IP are not as widely recognized by the public as traditional midstream segments of the semiconductor industry, such as wafer manufacturing and packaging/testing, they play an indispensable role in the entire industry ecosystem. In particular, EDA software essentially determines whether a chip can be designed, mass-produced, and ultimately meet core requirements such as high performance and low power consumption. It serves as an irreplaceable “infrastructure” in the modern chip design process.
The global EDA software market is currently highly concentrated. China remains heavily reliant on imports in this sector, with domestic alternatives still in their early stages and facing practical challenges such as limited technological accumulation and yet-to-be-established market trust. Against this backdrop, the SAMR’s exercise of its call-in power to require a notification and conduct a thorough assessment highlights the Chinese antitrust authority’s strategic focus and measured approach toward critical segments of the semiconductor industry, especially those exposed to acute “chokepoint” vulnerabilities.
Key Takeaways
Technology-intensive industries, exemplified by the semiconductor sector, will remain a key focus of antitrust regulation in China, with merger reviews and antitrust investigations serving as major enforcement tools. Particularly in sub-sectors with acute “chokepoint” vulnerabilities, where domestic alternatives have yet to establish effective competitive structures, the antitrust authority is likely to adopt more rigorous and forward-looking scrutiny to transactions involving industry-leading companies.
Focus on China’s supply security: addressing neighboring relationships and introducing behavioral remedies
In contrast to the antitrust reviews of this case in other jurisdictions, the SAMR not only identified the neighboring relationships between different types of EDA software, as well as between EDA software and design IP during the relevant market definition phase, but also paid close attention to the potential competitive impact of the transaction on these neighboring markets during the competitive analysis phase. The SAMR took into full consideration Ansys’ market position in several neighboring markets and the critical role of interoperability in the semiconductor design value chain, adopting a more cautious and stringent analytical approach. It ultimately concluded that the transaction would likely have the effect of eliminating or restricting competition in certain EDA software and design IP markets with neighboring relationships.
Based on this analytical framework, the SAMR designed and imposed a series of targeted behavioral remedies, key measures of which include requiring the parties to perform all existing contracts, maintain and, upon request, renew interoperability agreements with Chinese customers, prohibiting tying or discriminatory treatment, and supplying products to Chinese customers on FRAND (Fair, Reasonable, and Non-Discriminatory) terms.
These behavioral remedies, ranging from soft interoperability to contractual safeguards, are designed, on the one hand, to prevent “technology foreclosure” or “ecosystem lock-in” and to preserve space for innovation and access for market entry by domestic firms while China’s EDA industry is still in a nascent stage. On the other hand, they help ensure supply security, guaranteeing that the relevant products continue to serve the development of China’s downstream industries. This set of behavioral remedies, clearly aimed at strengthening competition in the domestic market, was not included in the conditional approval decisions of this case by other jurisdictions.
Key Takeaways
When designing remedy proposals for merger reviews, leading companies in technology-intensive industries need to pay particular attention to competition and supply chain security issues within the Chinese market. It is recommended that they proactively consider whether it is necessary to develop China-specific remedy packages and how to coordinate these measures with remedies implemented in other jurisdictions worldwide.
Prohibition of the Equity Acquisition of Shandong Huatai Pharmaceutical by Wuhan Yongtong
Wuhan Yongtong acquired a 50% equity interest in Shandong Huatai Pharmaceutical, thereby obtaining control over the latter. As the transaction did not meet the notification thresholds, the transaction parties did not submit it for review prior to implementation and completed the share transfer registration in March 2019. However, on January 3, 2025, the SAMR issued a written notice to Wuhan Yongtong requiring it to submit a merger notification for this concentration. Following its review, the SAMR concluded that the concentration would have the effect of eliminating or restricting competition in the market for papaverine hydrochloride injections within China and issued a decision to prohibit the concentration on July 22, 2025. The following aspects of this case are noteworthy:
Vertical integration in API sector causes competition concerns: transactions below notification thresholds are not exempt from review
In this case, neither the acquirer, Wuhan Yongtong, nor the target, Shandong Huatai Pharmaceutical, generated domestic revenue in China exceeding RMB800 million in 2017, and thus the transaction did not meet the notification thresholds. Consequently, the parties did not submit a filing before completing the share transfer registration. The failure to meet the notification threshold was partly attributable to the characteristics of the API sector, where the market size for individual drug types is often limited. As a result, the annual revenue of the relevant undertakings may fall short of the thresholds, meaning some transactions do not, in practice, require notification.
Nevertheless, as an upstream segment of the pharmaceutical industry chain, the API sector in China has long exhibited features such as high market concentration, significant entry barriers, and limited substitutability. Under these conditions, vertical integration with downstream drug product manufacturers presents a particularly acute risk of harming competition. Consequently, even where such transactions fall below the notification thresholds, they may still have a substantial impact on the market’s competitive landscape. In this case, the acquirer, Wuhan Yongtong, was the exclusive nationwide distributor for the papaverine hydrochloride API produced by Qinghai Pharmaceutical, already holding the dominant position in the relevant market, with downstream drug product manufacturers heavily dependent on its supply. The target, Shandong Huatai Pharmaceutical, was primarily engaged in the production of papaverine hydrochloride injections. Through the acquisition, Wuhan Yongtong could not only strengthen its dominant position in the upstream API market but also potentially extend its market power into the downstream formulation segment, resulting in a vertically integrated market structure. Therefore, although the transaction did not meet the notification thresholds, its potential to eliminate or restrict competition in the downstream market led the SAMR to call in this case and initiate a proactive review.
Key Takeaways
For companies with high market shares or even dominant market positions in sectors with strong monopolistic characteristics, such as APIs, it is essential to conduct a thorough assessment of whether a transaction may eliminate or restrict competition before its implementation, even if it falls below the notification thresholds. If the transaction is likely to adversely affect market competition, priority should be given to designing a deal structure that minimizes or mitigates competitive harm, or to evaluating the need for proactively notifying the SAMR of the below-threshold transaction to obtain clearance in advance. Taking such measures can help avoid the risk of retroactive enforcement action.
Significant downstream price surge as the “substantial” anti-competitive effect, leading to transaction prohibition
The SAMR not only identified potential market foreclosure concerns in the transaction structure, but also confirmed that the transaction had already caused significant anti-competitive effects in practice. This was particularly evident in the significant increase in the market share of the acquired downstream drug manufacturer and the sharp rise in injection prices, which substantially raised procurement costs for medical institutions and the financial burden on patients, while also depleting medical insurance funds. As a result, the SAMR decided to prohibit the transaction. As mentioned above, this case also marks the first case in the pharmaceutical industry of a prohibition decision requiring restoration to the pre-concentration state.
Furthermore, in another area of antitrust enforcement—cases involving the abuse of a dominant market position—APIs have long been a key focus. In recent years, antitrust authorities have repeatedly investigated and penalized API companies for leveraging their dominant positions to impose unfairly high prices, underscoring how the “price controllability” of key raw materials affects the fairness of the healthcare system. Therefore, in sectors closely tied to public welfare, such as pharmaceuticals, regulators often treat price and other factors that directly affect consumers’ vital interests as key considerations when assessing the competitive impact of a transaction.
Key Takeaways
In merger reviews, if a transaction could grant a company the exclusive control over key upstream resources, regulators typically pay close attention to the causal link between changes in the market supply structure and the corresponding movements in downstream product prices (as seen in this case with the structural change in API supply and the simultaneous surge in injection prices), and adopt this as a key basis for assessing whether the transaction has a substantial competitive impact.
Prohibiting the transaction and addressing the pre-existing exclusive distribution agreement
So far as the SAMR’s decision to prohibit the transaction and require restoration to the pre-concentration state is concerned, the decision not only required Wuhan Yongtong to “take a step back” by divesting the acquired equity in Shandong Huatai Pharmaceutical to an unaffiliated third party, thereby restoring the pre-concentration ownership structure, but also mandated “a step further” by requiring Wuhan Yongtong to terminate the exclusive API distribution agreement it had entered into with upstream suppliers two years prior to the concentration and to commit not to engage in any future concentrations of undertakings in the markets for papaverine hydrochloride API and papaverine hydrochloride injections.
These measures were designed not only to restore the pre-concentration market structure but also to curb Wuhan Yongtong’s ability to secure a dominant position by controlling the API supply channel, thereby preventing any future exclusionary or discriminatory monopolistic conduct in the relevant markets. We understand that the ultimate controller of the acquirer in this case was involved in a previously serious case of a horizontal monopoly agreement announced by the antitrust authority. Whether this case will serve as a broadly applicable precedent for transactions in the pharmaceutical sector remains to be seen in future antitrust enforcement.
Key Takeaways
For transactions that have already been implemented but are subsequently prohibited, if merely restoring the pre-concentration state is insufficient to eliminate the anti-competitive effect, the SAMR may even go further by imposing additional measures that are not directly tied to the transaction itself. For the companies involved, these requirements can force a reassessment and adjustment of their existing market strategies and development plans, potentially having unpredictable and far-reaching impacts on their ongoing business operations.
Compliance Recommendations
Based on the SAMR’s recent decisions on the two transactions that fell below the notification thresholds, we have summarized below the antitrust compliance recommendations to related industries:
Be cautious when implementing business arrangements with potential antitrust concerns, particularly in areas of enforcement priorities for China’s antitrust regulation. The SAMR’s use of its call-in power for transactions below the notification thresholds represents a shift from ex-post regulation (such as launching antitrust investigations into suspected monopolistic behavior) to ex-ante regulation. This reflects the antitrust authority’s concern over potential competition issues arising from transactions and its prudent regulatory stance in sectors crucial to national industrial development and public welfare, such as semiconductors and pharmaceuticals. The two decisions indicate that in technology-intensive sectors like semiconductors, China’s antitrust authorities focus on conducts that affect the stable supply, openness, and compatibility of technology.
When formulating global customer strategies, leading global technology companies should integrate the potential impact on Chinese customers and competition in the Chinese market into their compliance assessments. Companies should assess whether offering different commercial terms to customers outside China could be interpreted as discriminatory treatment against Chinese customers, potentially triggering antitrust scrutiny in China. Moreover, unjustified technology ecosystem foreclosure against Chinese customers or competitors, or imposing unreasonable transaction terms—such as excessive pricing, tying, or refusal to license or supply—may significantly increase the risk of being deemed unlawful in China. In the pharmaceutical sector, obtaining a monopoly position—whether through transactions, cooperation agreements, or other means—and subsequently implementing substantial price increase or refusing to supply to downstream companies in a way that disrupts the stable provision of drugs at reasonable prices, also carries a high risk of being considered unlawful.
Carefully assess your company’s market power in the relevant markets and the potential competitive harm a transaction may cause. In both merger reviews and antitrust investigations, China’s enforcement authorities are increasingly inclined to define relatively narrow relevant markets—whether for products, services, or technologies—to prudently evaluate the potential impact of a transaction or business arrangement across all affected markets. Companies should not underestimate potential antitrust risks simply because a product is in the early stages of R&D or market launch, or because its market share appears small within a broader category. When engaging in transactions or collaborations with competitors, upstream suppliers, or downstream customers, companies should carefully evaluate whether the relevant transaction, collaboration or the commercial terms involved could risk triggering an abuse of a dominant position or a notification obligation or a prohibition due to significant competition harm.
It is advisable to engage professional external counsel—and, where necessary, an economic analysis team—at an early stage to conduct a competitive assessment. This enables the design of transaction structures or commercial arrangements with minimal or relatively minor competition harm at lower cost, ensuring the smooth execution of the transaction or business cooperation without being hindered by antitrust regulatory scrutiny.
Take into account the unique competitive landscape of the Chinese market and design China-specific remedy packages at an early stage. For multinational corporations with advanced technologies, even if a transaction occurs outside China and does not meet China’s antitrust notification thresholds, they are recommended to still conduct a thorough, proactive assessment of the transaction’s impact on the Chinese market and determine whether a voluntary filing in China is warranted. In any case, a China-specific remedy package should be developed as early as possible to ensure that the transaction can obtain antitrust clearance in China smoothly. The case review of Synopsys’ acquisition of Ansys in China took over a year from initial submission to approval—the longest review period of all jurisdictions. Accordingly, when designing closing conditions, the time required for case review in China should be fully considered to avoid any adverse impact on the transaction from failing to meet the closing deadline.
Stakeholder companies could fully leverage antitrust complaint and litigation mechanisms to safeguard their legitimate rights and interests. As seen from the two cases above and other high-profile antitrust filing cases and investigations, antitrust compliance is not only a crucial means for companies to enhance economic benefit, but also an indispensable tool for companies to safeguard their legitimate interests in the business battlefield.
- For instance, Italy’s antitrust authority has invoked its call-in powers nine times in just two years, while jurisdictions like France and the Netherlands have initiated public consultations on legislation related to such powers. ↑
- According to Ansys’ 2023 Annual Report, the combined revenue of Ansys in Mainland China and Hong Kong SAR was US$111 million (approximately RMB785 million), which did not exceed the RMB800 million revenue notification threshold. See: https://investors.ansys.com/static-files/1fb38cf6-7360-4e51-ae68-6fcbc74e288a ↑
- Article 26(2): “Where a concentration of undertakings does not meet the notification thresholds prescribed by the State Council, but there is evidence that the concentration has or may have the effect of excluding or restricting competition, the antitrust authority of the State Council may require the undertakings to notify the concentration.” ↑